|
The s takes place arising from the existing electrons in the delocalized states. The Pauli exclusion principle states that no two electrons in the same atom can have the same set of quantum numbers. For the two electron in the orbital, the first three quantum number(n,l,$m_l$) are same, only the spin quantum number ($m_s$) differ. According to the Pauli exclusion principle, the electrons can be classified into state zero or state one 92. The salient features of the Pauli exclusion principle are as follows: In a given orbital only two electrons can occupy different quantum states.This can be summed up as : the maximum number of electrons in the shell with principal quantum number n is equal to $2n^2$. It can be also stated as only two electron can exists in the orbital and these electrons must have opposite spinģp -subshell can have maximum 6 electronsģd- subshell can have maximum 10 electrons.For example, if two electrons reside in the same orbital, then their n, l, and $m_l$ values are the same, therefore their $m_s$ must be different, and thus the electrons must have opposite half-integer spin projections of 1/2 and -1/2. As per this rule,it is impossible for two electrons of a poly-electron atom to have the same values of the four quantum numbers: n, the principal quantum number, l, the azimuthal quantum number, $m_l$, the magnetic quantum number, and $m_s$, the spin quantum number.According to this principle no two electrons can have same set of 4 quantum number.The number of electrons to be filled in various orbitals is restricted by the exclusion principle Le principe dexclusion de Pauli est responsable du fait que la matière ordinaire est stable et occupe un volume. The exclusion principle asserts that, in a system of identical particles, no two fermions (see below) can simultaneously occupy the same single-particle quantum. Paulis Exclusion Principle states that no two electrons in the same atom can have identical values for all four of their quantum numbers.This repulsive force is more than just an electrostatic repulsion between the electron clouds of the two atoms. 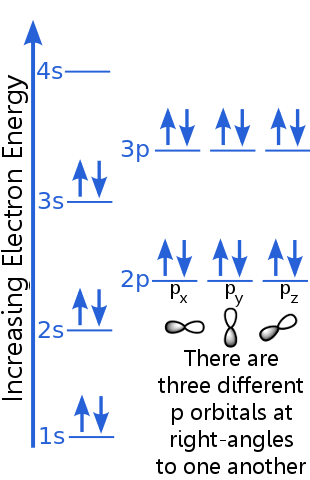
This principle was given by the Austrian scientist Wolfgang Pauli (1926) It is modeled above with two parameters C and a which can be adjusted to fit the data.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
